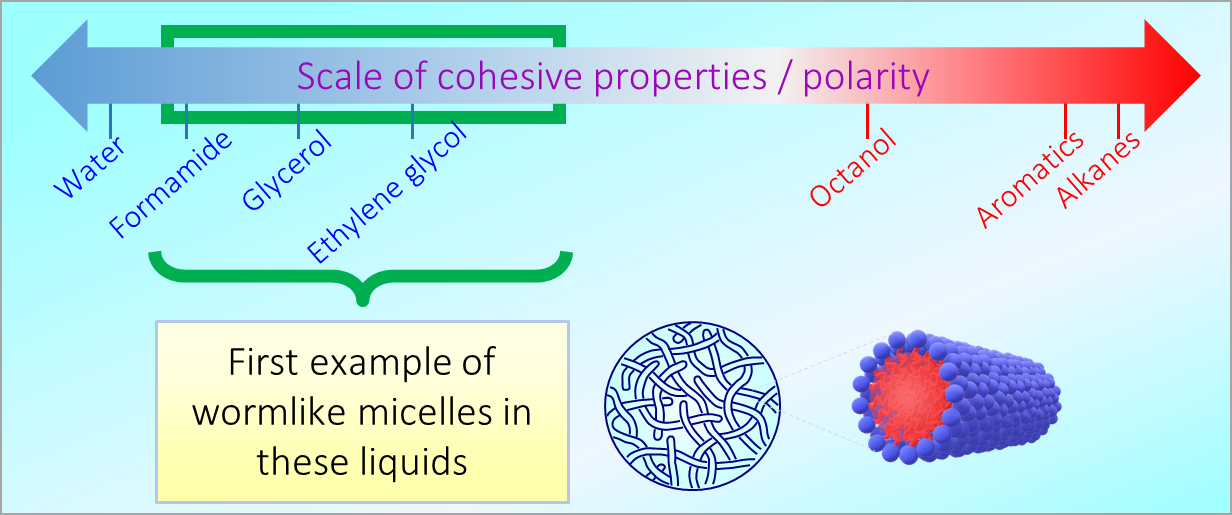
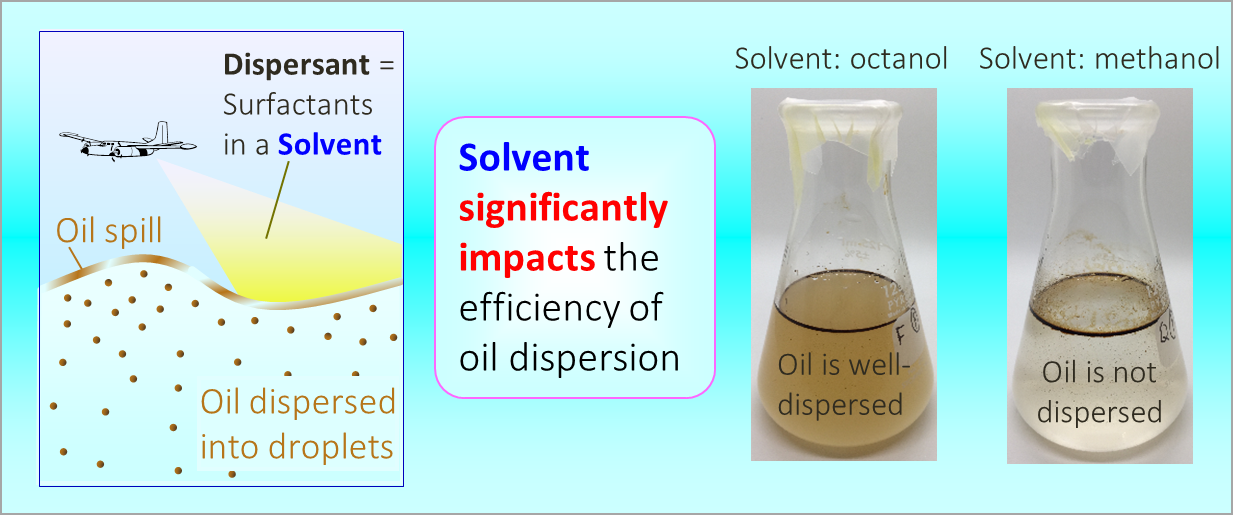
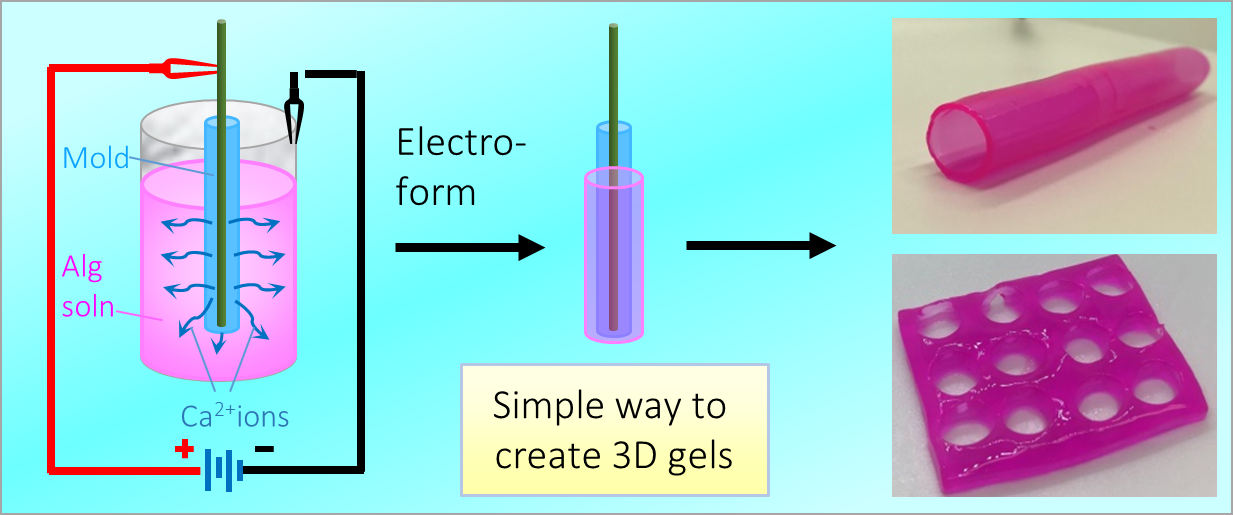
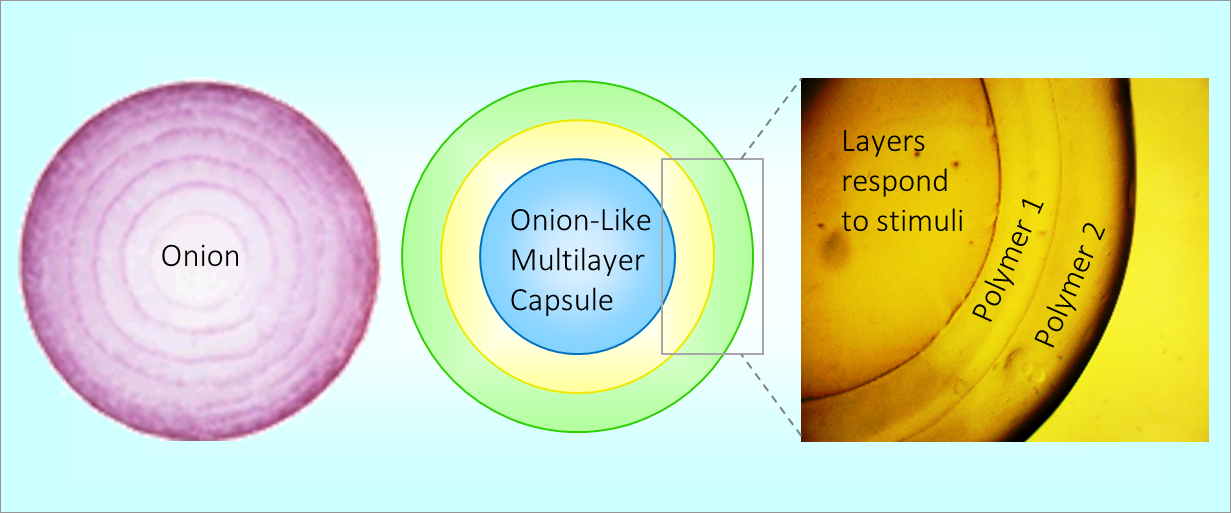
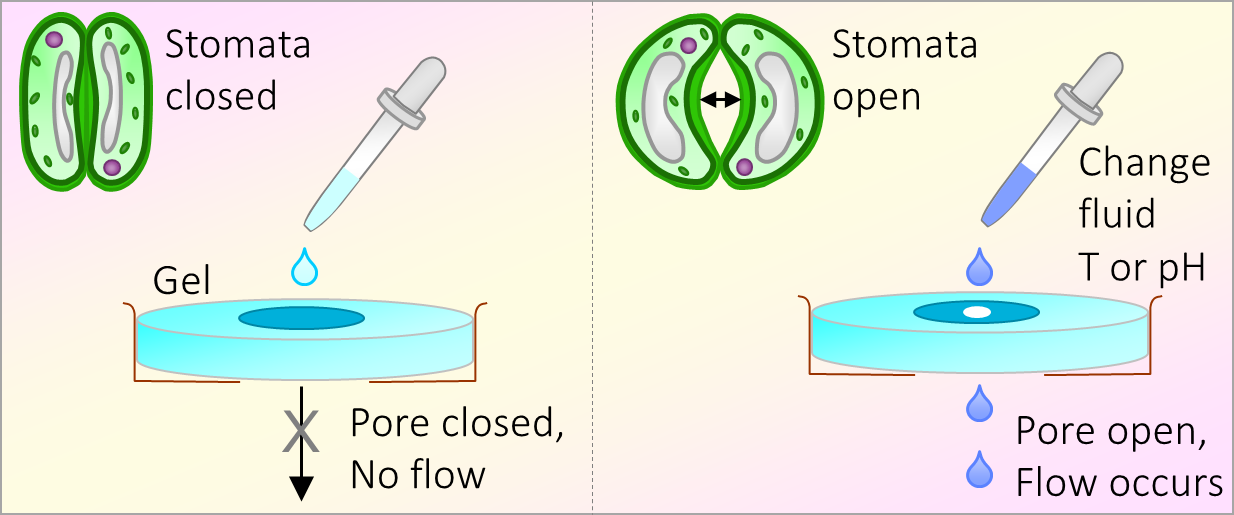
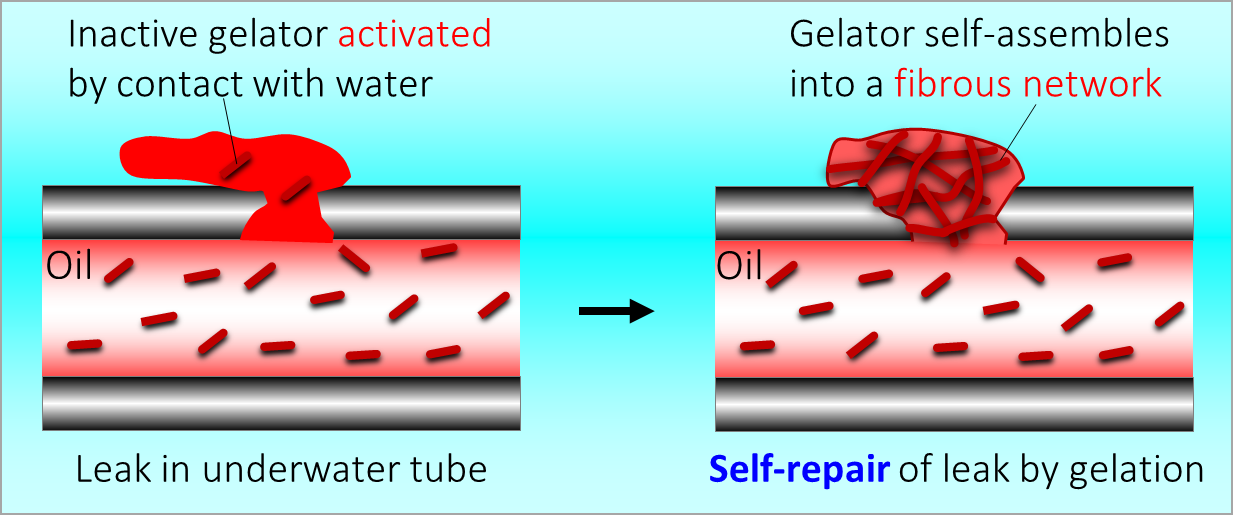
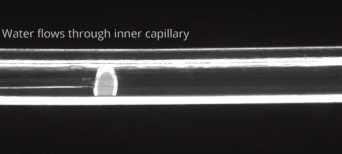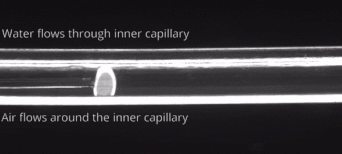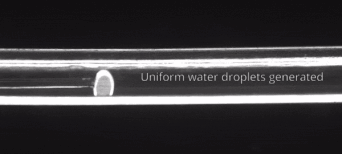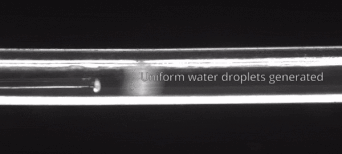
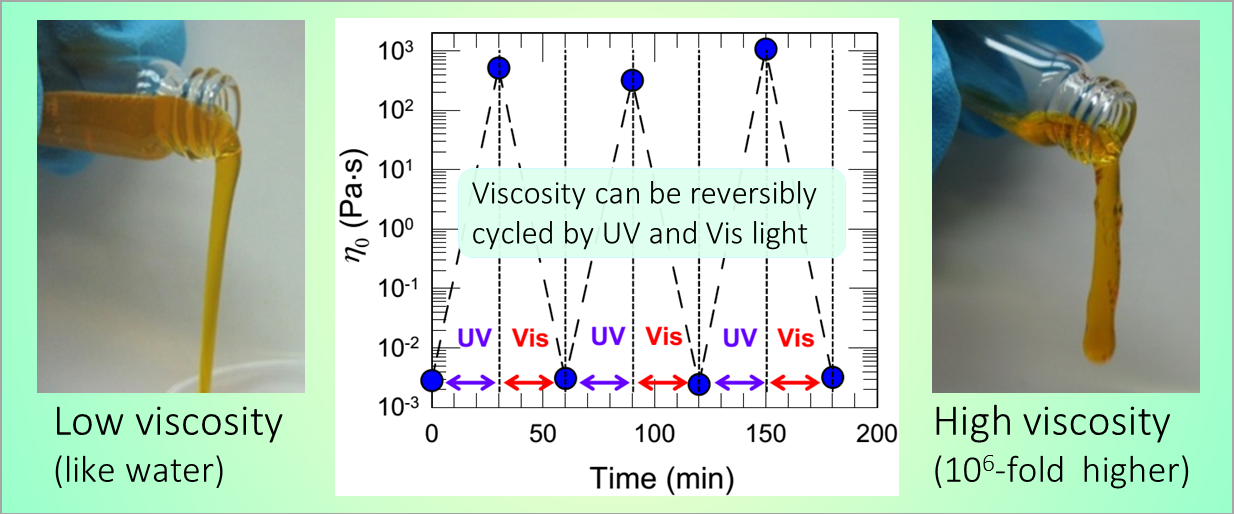
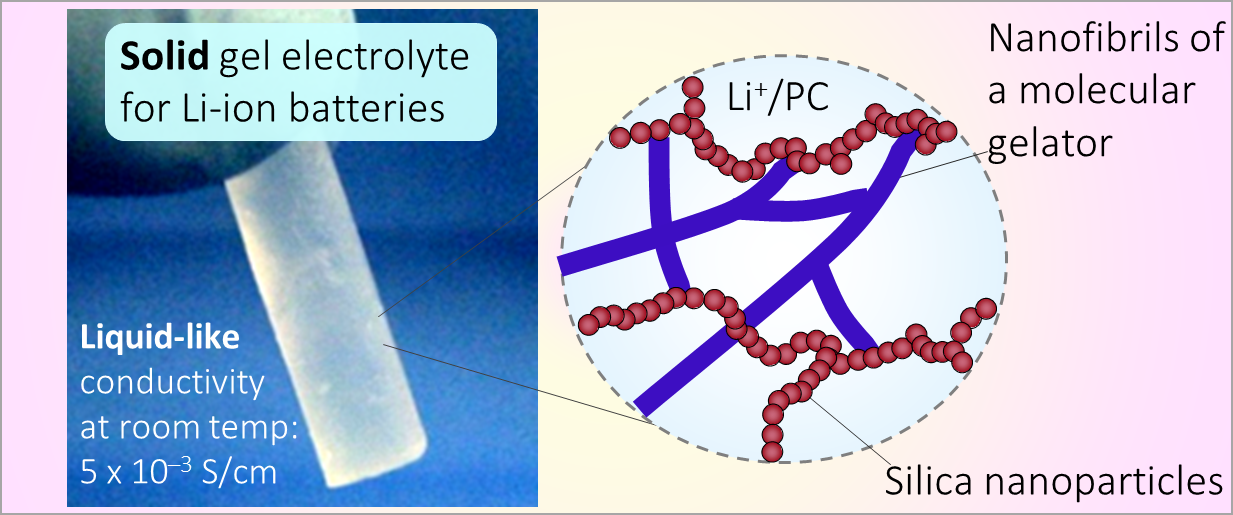
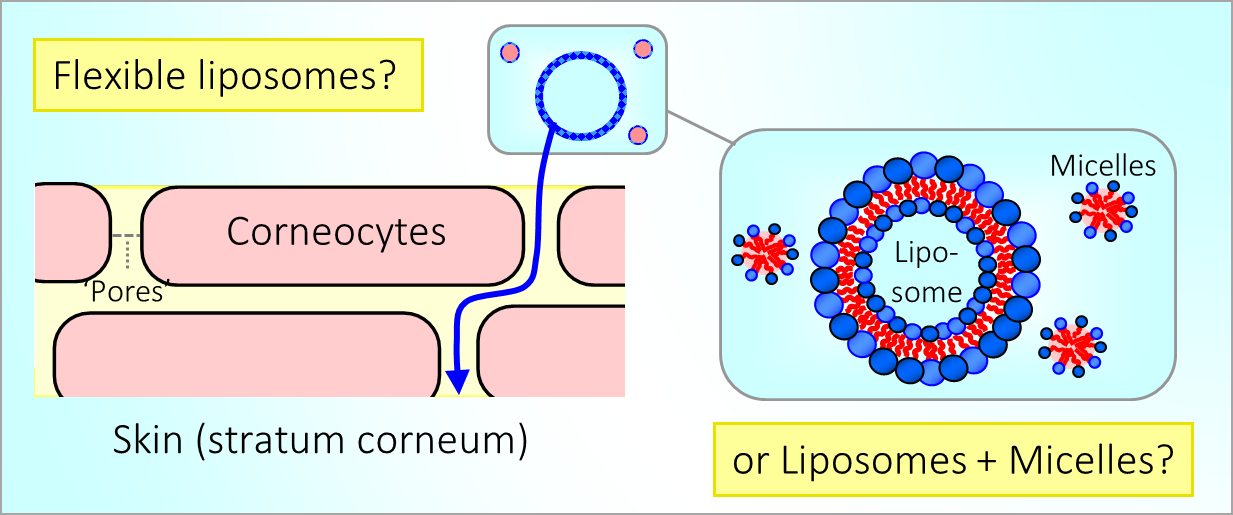
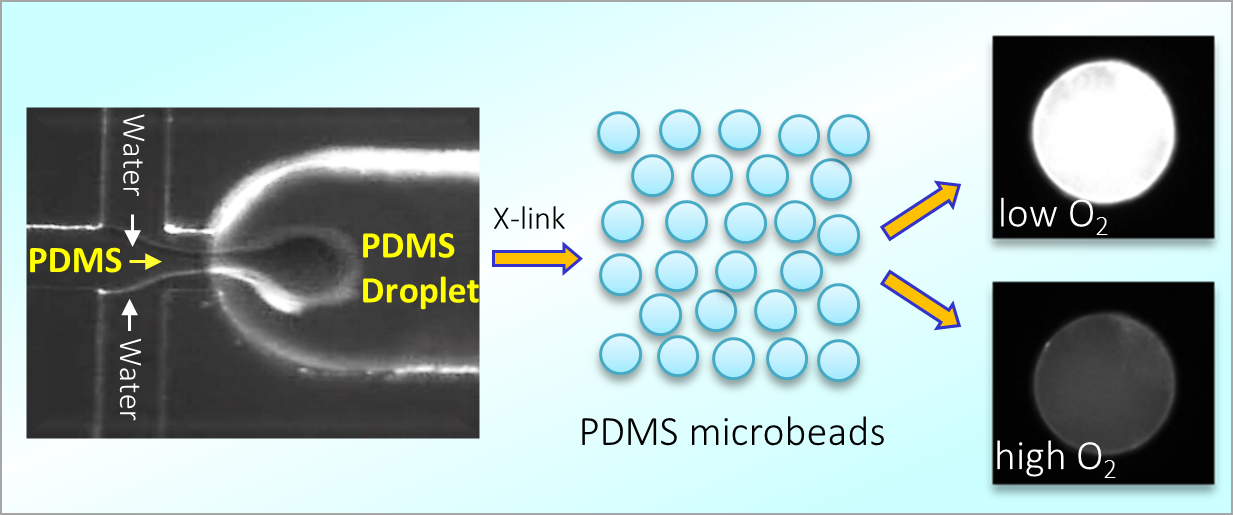
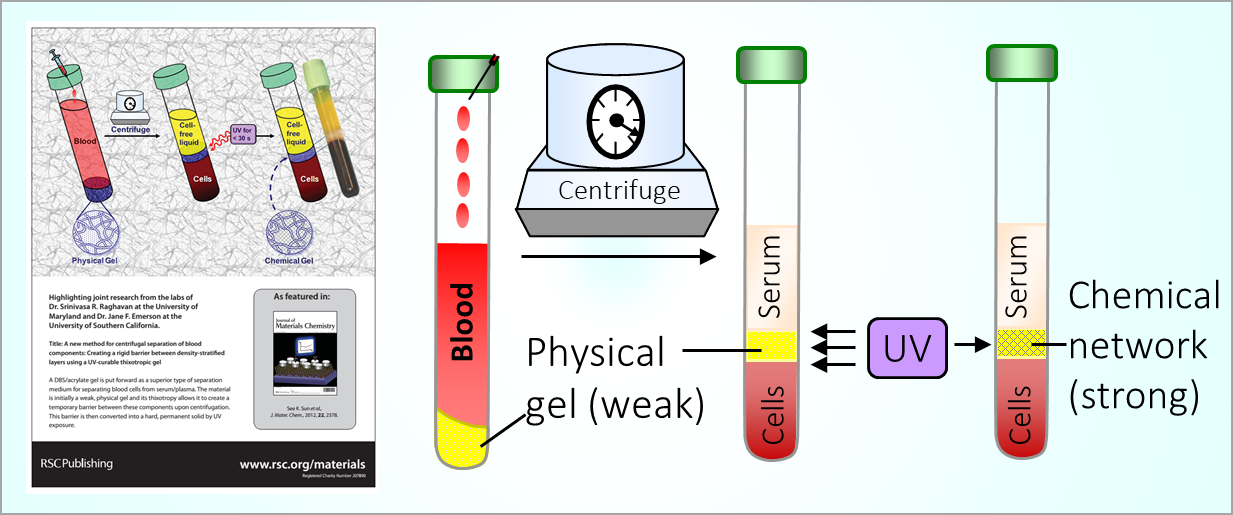
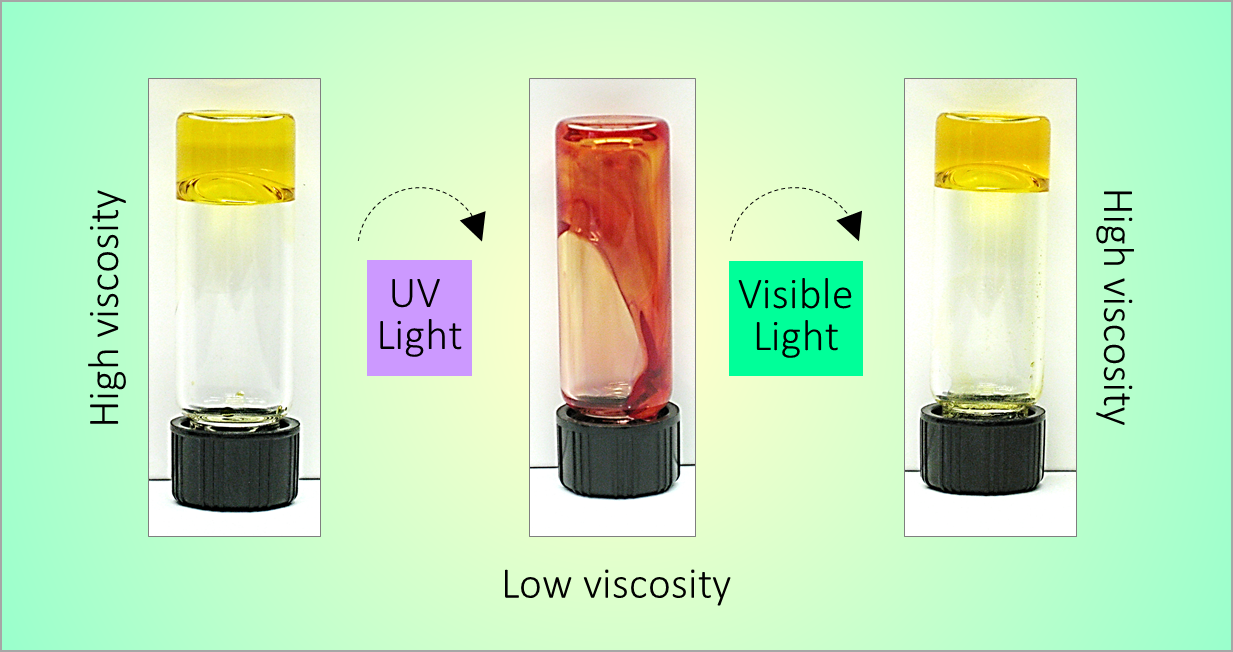
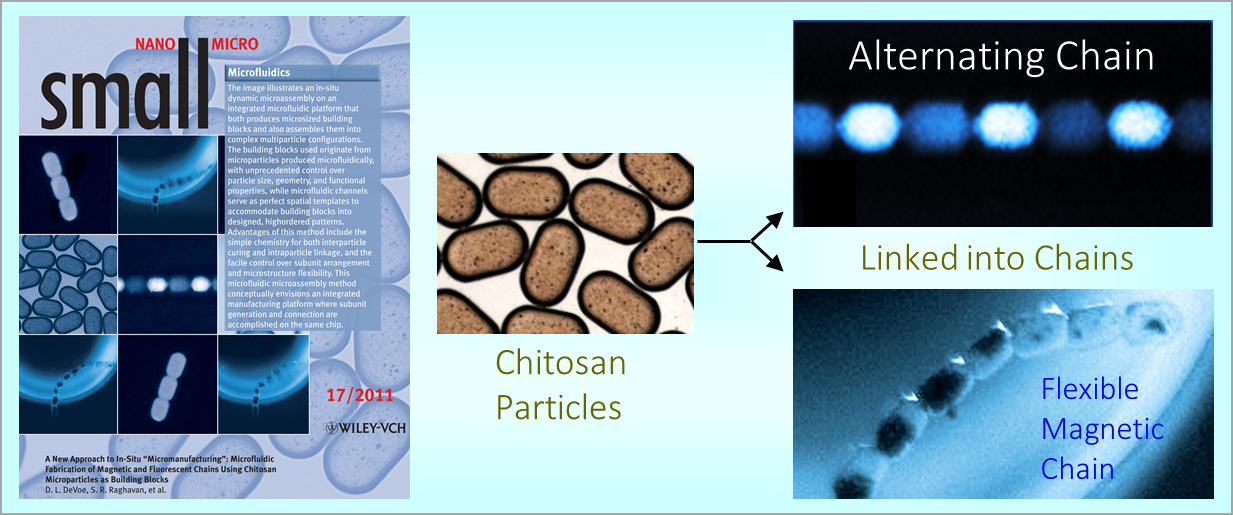
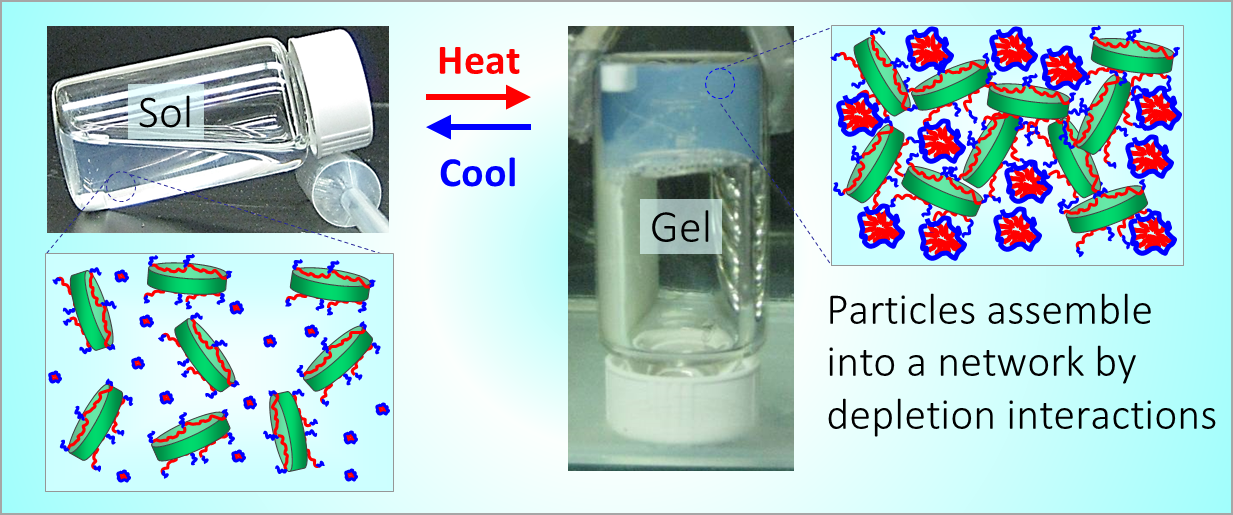
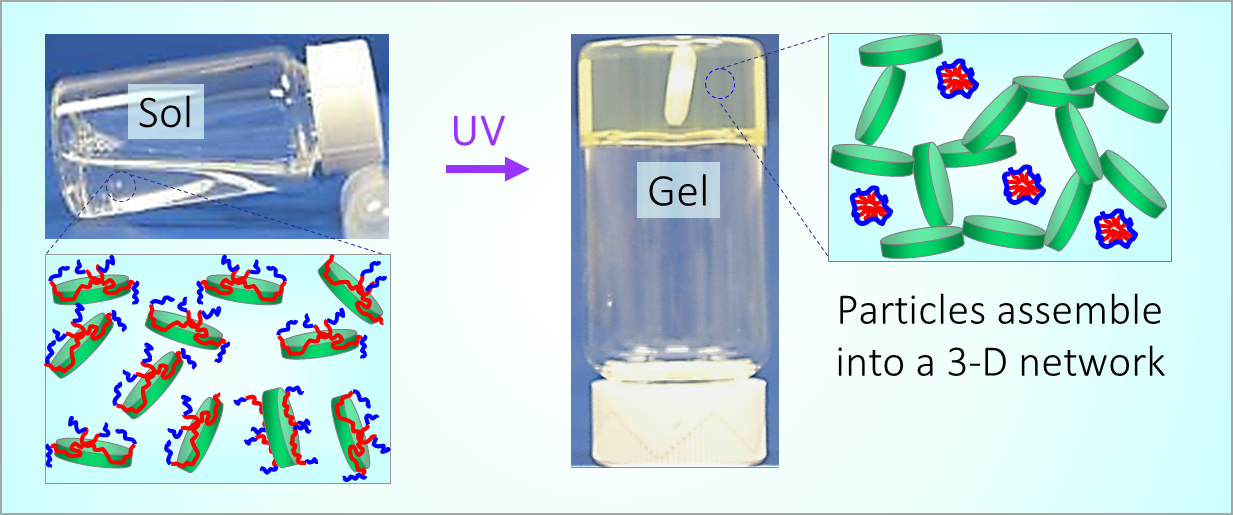
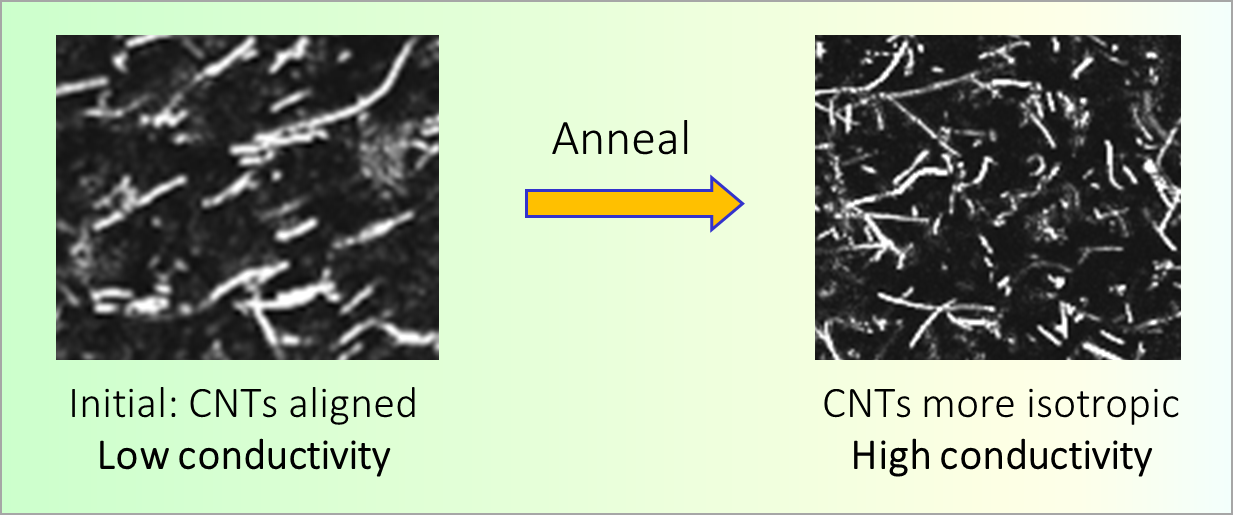
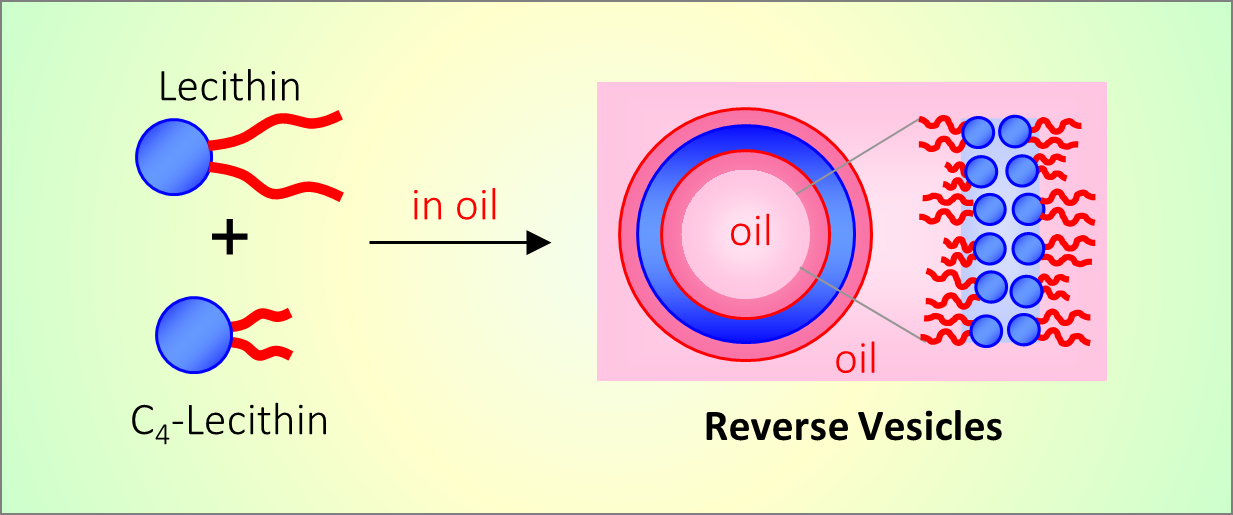
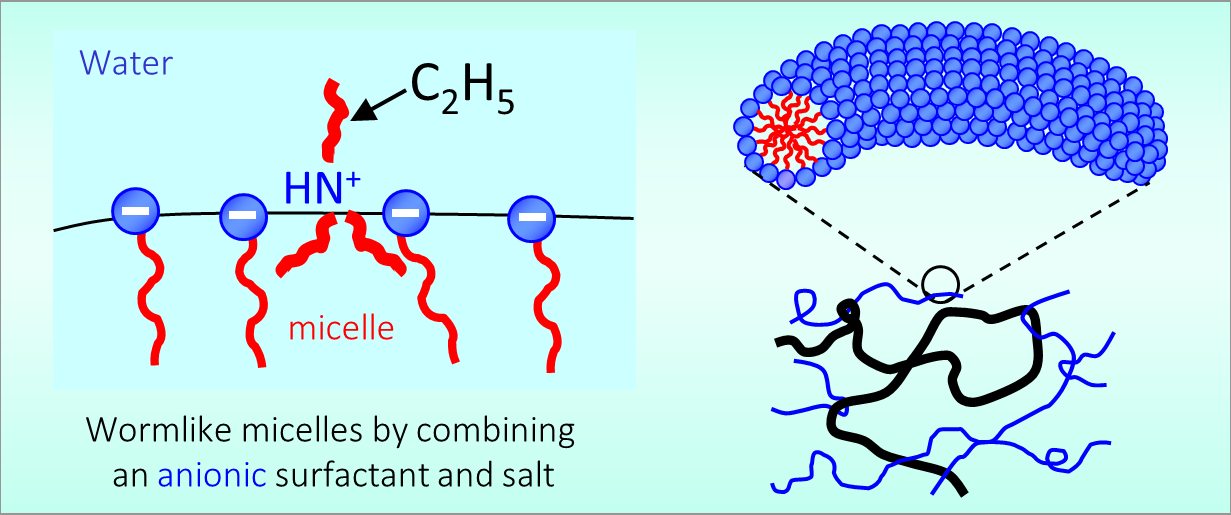
164. M. Z. Jora, E. Sabadini, S. R. Raghavan
2020
163. J. Zhang,..H. Choudhary, S. R. Raghavan,..C. Wang
“Water-in-salt” polymer electrolyte for Li-ion batteries.
Energy & Environmental Science, 9, 8357 (2020)
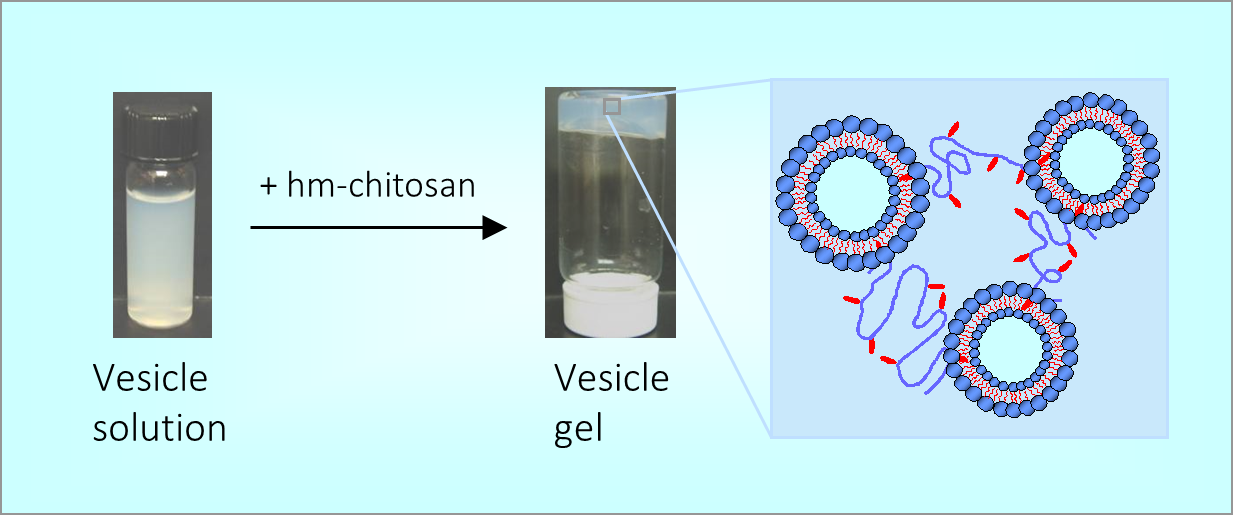
162. I. C. MacIntire, M. B. Dowling, S. R. Raghavan
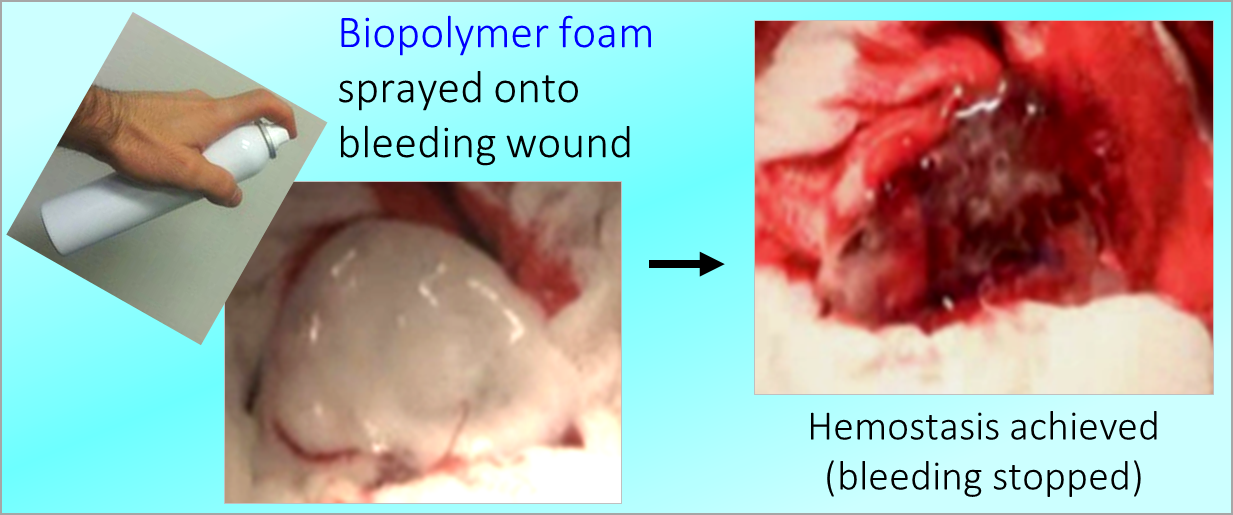
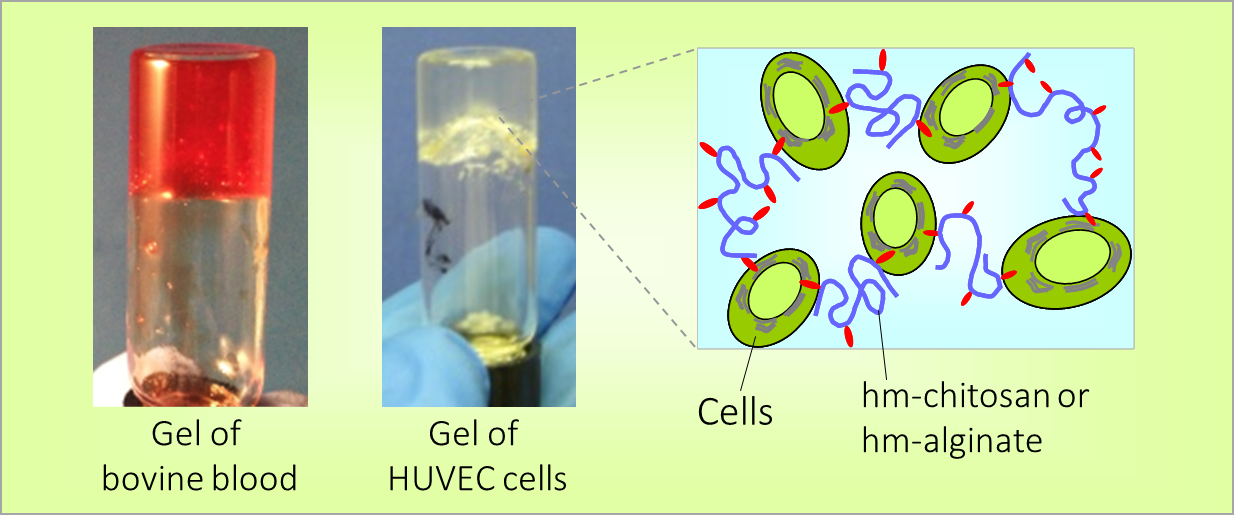
How do amphiphilic biopolymers gel blood? An investigation using optical microscopy.
Langmuir, 36, 8357 (2020)
161. B. R. Thompson, B. C. Zarket,…S. R. Raghavan
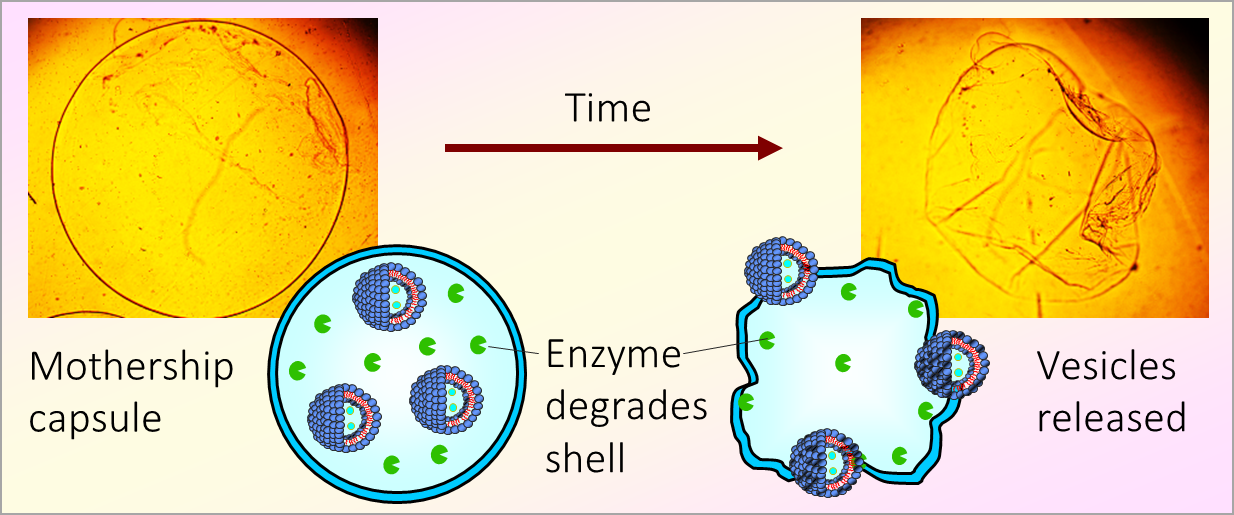
Liposomes entrapped in biopolymer hydrogels can spontaneously release into the external solution.
Langmuir, 36, 7268 (2020)
160. N. R. Agrawal, X. Yue, S. R. Raghavan
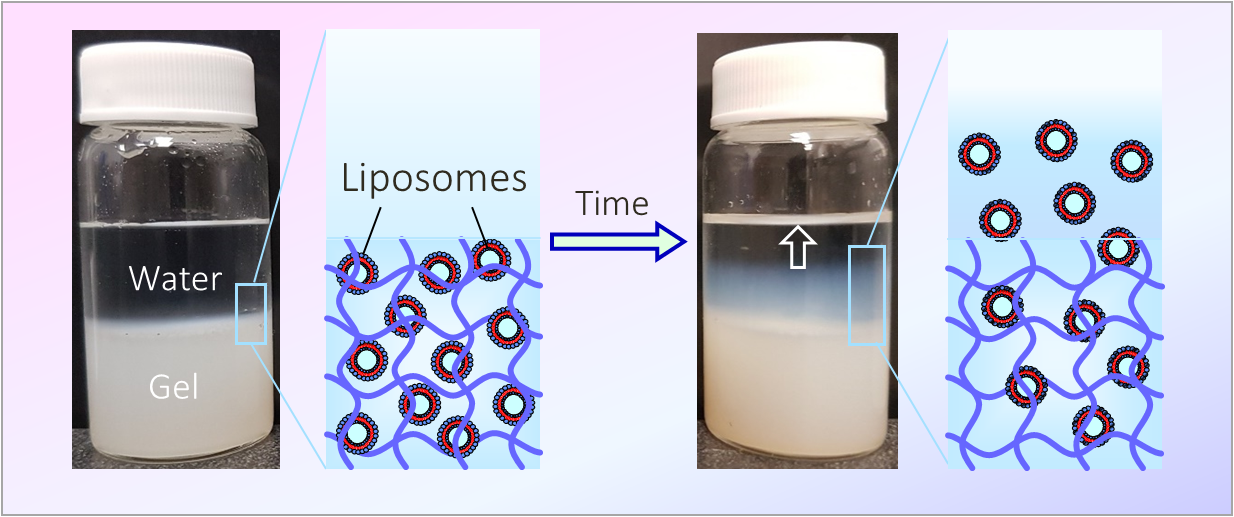
Liposomes are soft nanocontainers used to deliver drugs or cosmetics. They can be entrapped in gels of biopolymers like agar. We found, to our surprise, that entrapped liposomes can escape out of the gel network into water – possibly by squeezing through the pores in the network.
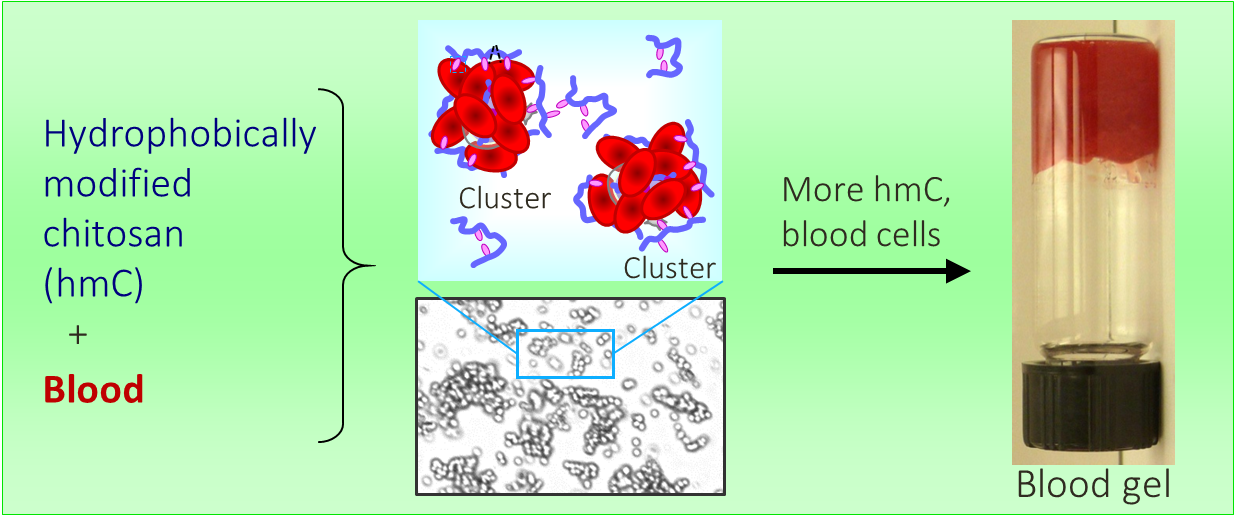
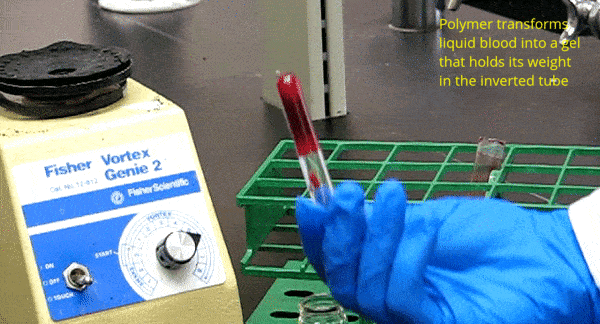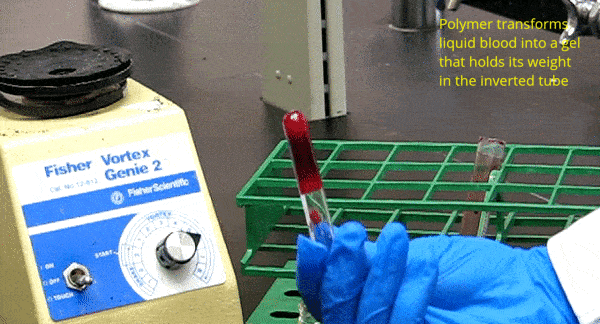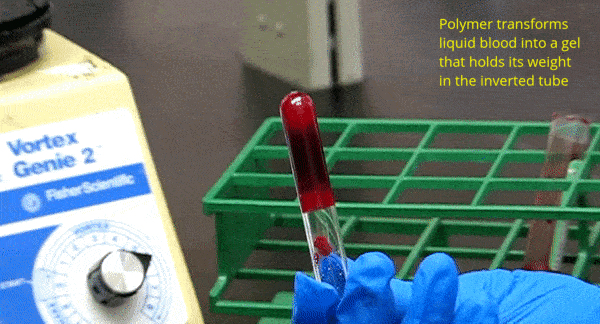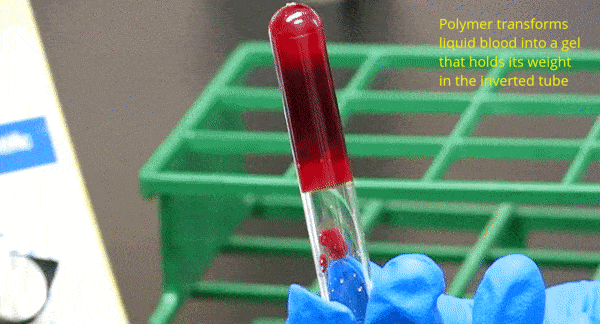
One of our key discoveries is the ability of amphiphilic biopolymers like hmC to convert liquid blood into a gel that retains its weight in an inverted vial. In this paper, we used optical microscopy to probe the underlying mechanism. We found that hmC induces blood cells to cluster, and these clusters connect into a network.